Chondrosarcoma
All information given here is only of a general nature, tumor therapy always belongs in the hands of an experienced oncologist!
Synonyms
Cartilage sarcoma, malignant chondroid tumor, enchondroma malignum, chondroblastic sarcoma, chondromyxoid sarcoma, chondroid sarcoma
English: chondroblastic sarcoma, chondrosarcoma
definition
Chondrosarcoma is a malignant tumor that is derived from cartilage cells.
In rare cases, a chondrosarcoma can appear in different places at the same time. In these cases one speaks of a chondrosarcomatosis.
After the osteosarcoma, the chondrosarcoma is the most common malignant (malignant) bone tumor.
frequency
With a share of 20%, chondrosarcoma is the second most common solid malignant bone tumor.
The peak of the disease in adulthood is between the ages of 30 and 50, but can in principle occur at any age.
localization
Chondrosarcoma occurs primarily in the following locations:
Frequencies
23% thigh
19% iliac bone
5% pubic bone
2% ischium
10% upper arm close to shoulder
5% shoulder blade
The chondrosarcoma is most common in the vicinity of the hip joint (Thigh and pool) localized (49%). The second most common location is the shoulder region with 15%.
root cause
The cause of the primary chondrosarcoma is not clear. Chondrosarcoma are derived from highly differentiated cartilage cells. The more differentiated a tumor is, that is, the more similar the tumor is to the original cell under the microscope, the more benign it behaves.
Secondary chondrosarcomas develop from benign chondromas. The malignant degeneration of an individual Enchondroma is altogether unlikely.
The risk of degeneration increases with the number of enchondromas present. The risk of degeneration of a single enchondroma is estimated at around 1%.
However, there is a high risk of degeneration in enchondromatosis with or without the presence of Ollier's disease and in Maffucci syndrome. If there are many osteochondromas, the risk of degeneration is estimated to be significantly higher at around 10%.
metastasis
In most cases, chondrosarcoma is a tumor with a high degree of differentiation (see above).
The transitions from benign cartilage cells to malignant tumors are fluid and often difficult to differentiate.
The decrease in differentiation (similarity of the tumor tissue with the original tissue) is accompanied by an increase in malignancy. In the same way, the probability of metastases increases and the prognosis worsens. Differentiation is therefore an important prognostic factor.
Chondrosarcoma metastasize primarily hematogenously to the lungs.
classification
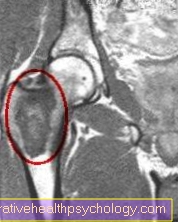
There are many classifications that describe different subtypes. The differentiation is essentially based on the fine tissue examination under the microscope.
Primary chondrosarcoma:
- Chondrosarcoma (conventional)
- Dedifferentiated chondrosarcoma
- Juxtacortical (periosteal) chondrosarcoma
- Mesenchymal chondrosarcoma
- Clear cell chondrosarcoma
- Malignant chondroblastoma
- Secondary chondrosarcoma
differentiation
The likelihood of malignancy increases, especially when a tumor occurs close to the trunk, i.e. not on the arms and legs.
Chondrosarcomas that occur close to the trunk usually have different areas. This means that there are areas in which the tumor is "still benign" and has already reached malignancy in other areas. Therefore, the entire tumor must always be examined under the microscope.
Furthermore, all available sources of information must be compiled in order to exclude a tumor (examination findings, X-ray and other imaging procedures, tissue examination).
The following principles are valid:
- Larger tumors close to the trunk or tumors that change on the X-ray should be completely removed. If a malignancy turns out later, the cut edges should be removed again with an appropriate safety margin.
- Chondrosarcomas on the fingers and feet tend to be benign behavior, even if they show all signs of malignancy under the microscope.
Diagnosis
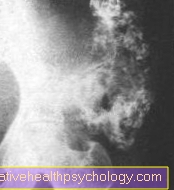
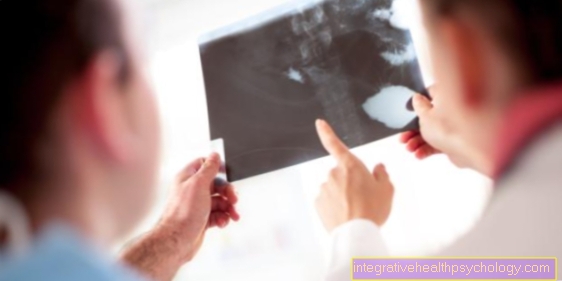
Imaging diagnostics:
- X-ray image in two planes of the suspect area
- Sonography (ultrasound) of the tumor (if it should not consist of bone or cartilage)
- Blood test:
- Blood count
- ESR (sedimentation rate)
- CRP (C-reactive protein)
- Electrolytes (when the bone is attacked, the calcium in the blood increases)
- Alkaline phosphatase (aP) and bone-specific aP: increased in bone-dissolving (osteolytic) processes
- Prostate-specific antigen (PSA): increased in prostate approx
Acid phosphatase (sP): increased in prostate ca - Uric acid (HRS): increased with high cell turnover (very active tumor)
Iron: decreased in tumors - Total protein: decreased in consuming processes
- Protein electrophoresis, immune fixation (special tests)
- Urine status: paraproteins - evidence of plasmacytoma
Special tumor diagnostics:
- Local diagnostics (= apparatus-based examination of the local tumor):
- MRI:
With the MRI, the spread of the tumor into neighboring structures such as Muscle tissue, nerves and vessels are clarified. The use of contract agents can improve the distinction between benign and malignant tissue.
Read more on the topic: MRI with gadolinium contrast agent - CT:
The CT particularly provides information about the bone involvement of the tumor - PET (positron emission tomography):
(Value, however, not yet sufficiently validated)
- MRI:
- Locoregional diagnostics (= search for lymph node metastases which rarely occur in chondrosarcoma):
- Sonography (ultrasound)
- possibly CT
- MRI if necessary
- Extra regional diagnostics:
- Examination of organs that are particularly frequently affected by chondrosarcoma metastases: - primarily the lungs, liver and adrenal glands.
- Systemic diagnostics (= spread diagnostics and general tumor search):
- Skeletal scintigraphy (3-phase scintigraphy)
- PET (positron emission tomography; value not yet sufficiently validated)
- Special tumor laboratory diagnostics
- Immunoelectrophoresis: evidence of plasmacytoma if monoclonal antibodies are detected
- Hemoccult test (detection of blood in the stool)
- Tumor markers (e.g. NSE = neuron-specific enolase in Ewing's sarcoma)
Tumor markers have no diagnostic value in chondrosarcoma, as there are no reliable tumor markers that indicate a chondrosarcoma.
Biopsy:
If the benign or malignant nature of the tumor cannot be clearly determined, a sample (biopsy) of the suspicious area can be carried out so that it can then be examined in terms of tissue.
It should be noted, however, that this sampling also causes a so-called scattered metastasis, in which the tumor is detached from its compound
Read more about this topic here: biopsy
Tumor aftercare
Recommendations:
- In year 1 and 2: every 3 months clinical examination, local X-ray control, laboratory, chest CT, whole-body skeletal scintigraphy, every 6 months local MRI
- In years 3 to 5: every 6 months clinical examination, local X-ray control, laboratory, chest CT, whole-body skeletal scintigraphy, local MRI every 12 months
- From year 6: every 12 months clinical examination, local X-ray control, laboratory, chest X-ray, in case of doubt whole-body skeletal scintigraphy and local MRI
forecast
The prognosis depends on the degree of tissue differentiation and the possibility of radical surgery. If the degree of differentiation is high and "radical" surgery is possible, the probability of survival for 5 years is approx. 90%.
A renewed tumor growth can still occur after more than 10 years.











.jpg)