Trypsin
introduction
Trypsin is an enzyme that is produced in the pancreas and is of great importance for human digestion. It activates other digestive enzymes from the pancreas in the intestine, which in turn further break down proteins that are ingested with food. These can then be absorbed in the further course of the passage through the intestine. Thus, as an activator for various digestive enzymes, trypsin is important for the absorption of proteins.
Tasks and function - how does trypsin work?
Trypsin is produced in the pancreas (pancreas) and, as an enzyme, is responsible for activating other digestive enzymes from the pancreas. These are mainly chymotrypsin, elastase and carboxypeptidase. Trypsin thus triggers an activation cascade that is of crucial importance for the digestion of proteins from food. A deficiency therefore leads to serious disorders in the body and especially in the large intestine to putrefaction.
also read:
- Chymotrypsin
- Carboxypeptidase
The enzymes that trypsin activate can in turn chop up proteins from food and break them down into so-called oligopeptides. These can then in turn be more easily absorbed in the small intestine and transported to various parts of the body via the circulation. The body needs the protein components from food primarily in order to build its own proteins from them. These are used, for example, for the body structure in the form of skin, connective tissue, muscles or bones. The body also needs proteins as hormones, antibodies for the immune system or to transport oxygen.
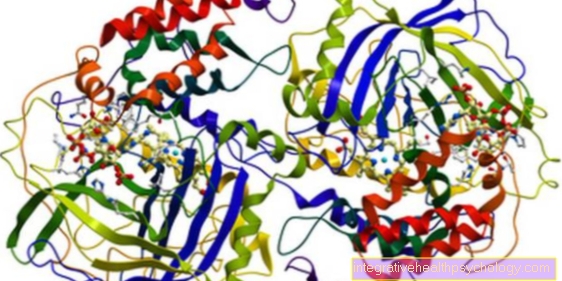
As an enzyme, trypsin has specific properties in terms of the way it breaks down proteins. It is a so-called endopeptidase, which means that it splits peptide bonds, i.e. the bonds between individual amino acids (which make up a peptide or protein) within the protein.
In addition to its main function in digestion, trypsin can also reduce pain that occurs, for example, in the context of arthritis.
It also plays a role in the breakdown of various complexes in autoimmune diseases and can prevent the excessive accumulation of platelets. The latter happens because trypsin activates other enzymes, such as plasmin, which in turn dissolve fibrin and thus loosen the adhering network of blood platelets
What is trypsinogen?
Trypsin is an enzyme that tends to break down other proteins. It is therefore important that this function does not come into effect at the place of manufacture, i.e. the pancreas. To prevent this, trypsin is produced in an inactive precursor. This precursor is also known as the proenzyme and, in the case of trypsin, is the trypsinogen.
Read more about the topic here: Trypsinogen
When eating, the inactive precursor trypsinogen is released from the pancreas and activated in the small intestine. This is done by another enzyme called enteropeptidase. Parts of the trypsinogen are split off, creating the active form trypsin.
When diagnosing some diseases, such as cystic fibrosis, it is important to measure the trypsin level in the human body. For this purpose, the amount of trypsinogen in the body can also be determined, because this is directly related to the amount of available trypsin.
You might also be interested in: Cystic fibrosis
What is antitrypsin?
Antitrypsin is a so-called protease inhibitor. This means that antitrypsin is able to prevent trypsin from doing its job and breaking down proteins. This is especially important for breaking down proteins in the blood.
Learn more about: Alpha-1 antitrypsin
Antitrypsin is usually referred to as alpha-1-antitrypsin because it can be detected in a detection method (protein electrophoresis) in the so-called alpha-1 fraction. It is largely made in the liver and is important in preventing excessive inflammation in the body by blocking various enzymes such as trypsin, plasmin, or thrombin. In the event of a deficiency, the liver and lungs are seriously impaired.
You might also be interested in: Alpha-1 antitrypsin deficiency
Trypsin inhibitors
Trypsin inhibitors are peptides that prevent trypsin from developing its effect in the intestine or limit it. Trypsin is blocked and cannot do its job as an activator of other digestive enzymes in the intestine.
Trypsin inhibitors are found in a variety of foods. A well-known representative are soybeans, which contain trypsin inhibitors in their raw form. When consuming raw soybeans, protein digestion in the intestine can be disrupted. To prevent this, the soybeans should be cooked before consumption, as this inactivates the trypsin inhibitors. In the case of soybean flour, it should be ensured that it has gone through a roasting process, as this also renders the trypsin inhibitors harmless. Otherwise, digestive problems could arise.
Read on below: Digestive problems
Laboratory values of trypsin
Standard value
A blood sample is required to determine the trypsin level. From this, the amount of trypsin present can be determined in a medical laboratory.
In a healthy person, the value is between 10 and 57 μg per liter of blood.
As a rule, the trypsin level is determined if there is a suspicion that an acute inflammation of the pancreas, i.e. pancreatitis, may be present. For this, however, other parameters such as the elastase value should also be determined.
Trypsin increased
If the level of trypsin in the blood is increased, this means that either an excessive amount of the digestive enzyme is released from the pancreas or too little trypsin is excreted from the body after its function has been carried out.
Excessive secretion of trypsin can be caused by an acute inflammation of the pancreas, i.e. pancreatitis. However, it can also be a relapse of a chronic, i.e. long-standing, pancreatitis. Furthermore, a tumor of the pancreas or a cystic pancreatic fibrosis in the context of cystic fibrosis are possible causes. If one of these diseases is suspected, however, further parameters of the pancreas should be assessed for clarification.
Another possible cause of the increase in trypsin levels is kidney failure. This disease should also be further clarified by a doctor if it is suspected.
Find out more at: Inflammation of the pancreas
Trypsin deficiency
Since trypsin is a crucial part of the activation cascade for digestive enzymes in the intestine, a deficiency leads to inadequate digestion and absorption of proteins from food.
The lack of absorption of food proteins leads to weight loss and deficiency symptoms due to a lack of proteins in the human body. In addition, there is a so-called intestinal putrefaction with an accumulation of nitrogen-containing compounds. This can also be recognized by meat fibers in the stool.
At what pH value does trypsin work best?
Trypsin, like most other digestive enzymes, can only work properly at a certain pH. The optimal pH range for trypsin is between 7 and 8 and thus corresponds to the pH range in a healthy person in the small intestine. If this range changes, trypsin can no longer work adequately and this can lead to a disruption of the absorption of proteins from food.
Find out about other enzymes and what they do: Role of enzymes in the human body
Production of trypsin
Trypsin, like many other digestive enzymes, is made in the pancreas, or more precisely the exocrine part of the pancreas. There it is first released as a proenzyme trypsinogen and then converted into an active form in the small intestine by the enzyme entereopeptidase, which in turn can activate further digestive enzymes.
Recommendations from the editorial team
You might also be interested in:
- Inflammation of the pancreas
- Digestive problems
- Alpha-1 antitrypsin deficiency
- Chymotrypsin
- Role of enzymes in the human body
- Alpha-glucosidase